 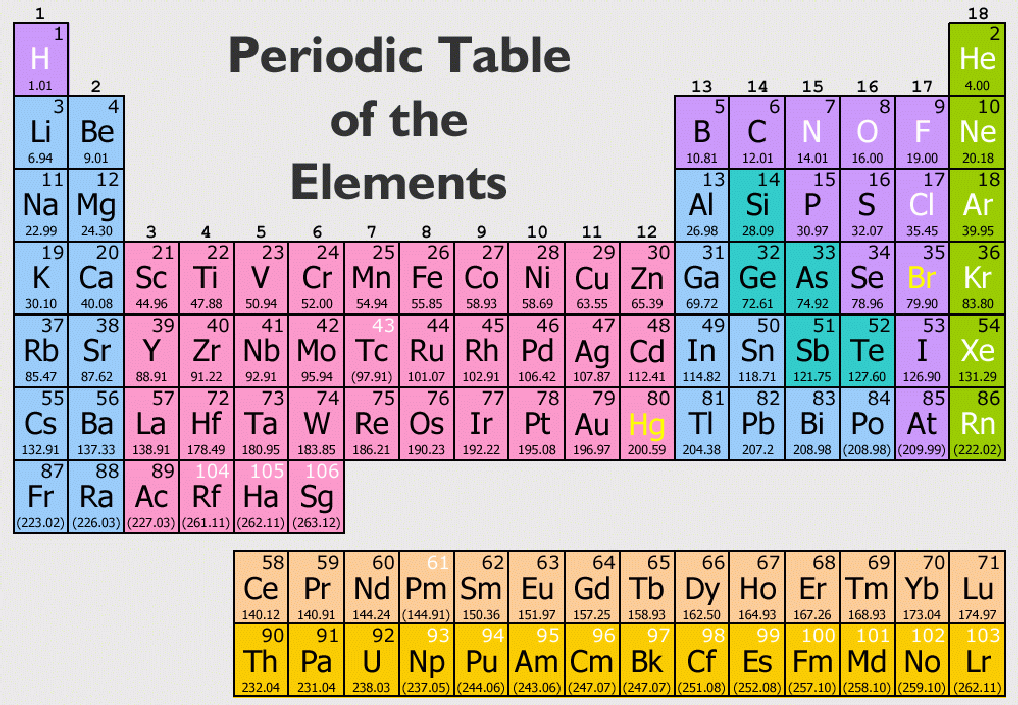 
Transplutonium elements – Elements with atomic number greater than 94.Transactinide elements – Elements after the actinides (atomic number greater than 103).Superactinides – Hypothetical series of elements 121 to 157, which includes a predicted "g-block" of the periodic table.Precious metals – Variously-defined group of non-radioactive metals of high economical value.Many other names have been used for this set, and its borders are not agreed on. Post-transition metals – The metals coming after the transition metals.Platinum group – Ru, Rh, Pd, Os, Ir, Pt.Non-ferrous metals - Metals or alloys that do not contain iron in appreciable amounts.Usually includes Ag, Au, and the platinum-group metals. 
Noble metals – Variously-defined group of metals that are generally resistant to corrosion.Native metals – Metals that occur pure in nature, including the noble metals and others such as Sn and Pb.Minor actinides – Actinides found in significant quantities in nuclear fuel, other than U and Pu: Np, Am, Cm.Heavy atom – term used in computational chemistry to refer to any element other than hydrogen and helium.Heavy metals – Variously-defined group of metals, on the base of their density, atomic number, or toxicity.Earth metal – Old historic term, usually referred to the metals of groups 3 and 13, although sometimes others such as beryllium and chromium are included as well.Coinage metals – Various metals used to mint coins, primarily the group 11 elements Cu, Ag, and Au.Metalloids – Variously-defined group of elements with properties intermediate between metals and nonmetals.These sets usually do not aim to cover the whole periodic table (as for example period does), and often overlap or have boundaries that differ between authors. Many other names for sets of elements are in common use others have been used throughout history. These two to three classes are commonly marked by differing background colors in the periodic table. Elements in the vicinity of where the metals and nonmetals meet are sometime classified as metalloids or an equivalent term. ‡ Lanthanoids and actinoids are sometimes referred to as lanthanides and actinides respectively Metallicity-based Īnother common classification is by degree of metallic or nonmetallic behaviour and characteristics. * Transition elements are sometimes referred to as transition metals † Although the heavier elements of groups 15 (Mc), 16 (Lv), 17 (Ts) and 18 (Og) have been notionally assigned to the indicated groups their chemical properties have not yet been experimentally confirmed. The elements of group 18: He, Ne, Ar, Kr, Xe, Rn †Įlements 57–71: La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, LuĮlements 89–103: Ac, Th, Pa, U, Np, Pu, Am, Cm, Bk, Cf, Es, Fm, Md, No, LrĮlements in groups 1–2 or 13–18, excluding hydrogen The elements of group 17: F, Cl, Br, I, At † The elements of group 16: O, S, Se, Te, Po † The elements of group 15: N, P, As, Sb, Bi † The metals of group 2: Be, Mg, Ca, Sr, Ba, RaĮlements in groups 3 to 11 or 3 to 12 (the latter making up the d-block) The metals of group 1: Li, Na, K, Rb, Cs, Fr The following collective names are recommended or noted by IUPAC: Many of these sets are formally recognized by the standards body IUPAC. Amongst this diversity, scientists have found it useful to apply names for various sets of elements that have similar properties, to varying degrees. There are currently 118 known chemical elements with a wide range of physical and chemical properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
